Directly from the manufacturer
Free shipping
Fast delivery
Secure payment
Directly from the manufacturer
EMS vs. BMS
The Compliance Gap Hiding in Your Building Management System

Why do I need a separate EMS when my BMS already monitors temperature? If you’ve asked this question, you’re not alone. This is a common inquiry from operations and compliance teams within a variety of pharmaceutical organizations, especially when they seek to streamline infrastructure and ensure compliance.
While a Building Management System (BMS) is vital for facility performance, it is not designed to meet the stringent GMP compliance and data integrity requirements of the pharmaceutical industry. The most advanced and efficient strategy is one that integrates both systems.
EMS vs. BMS: A Quick Comparison
The core difference between the two systems can be summarized by their distinct roles and purposes.
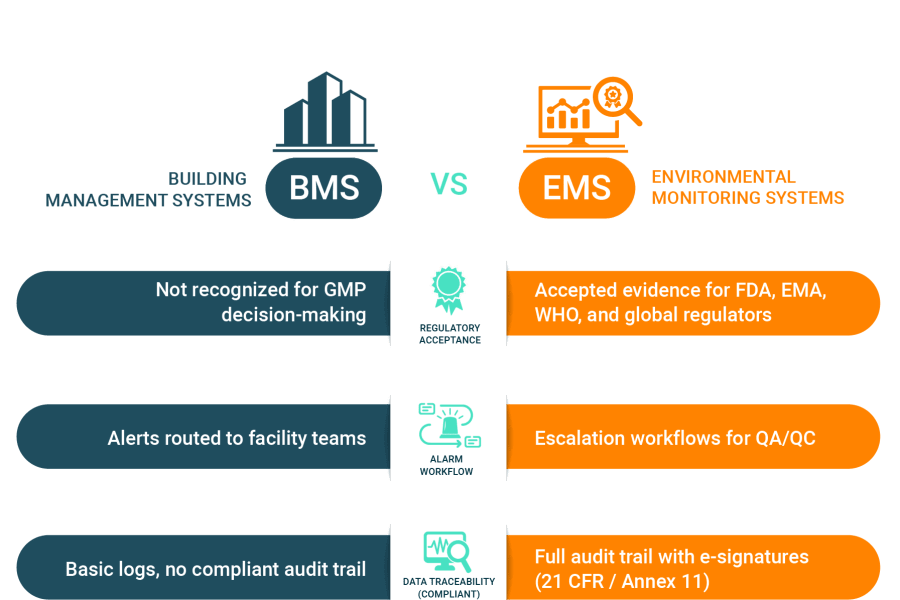
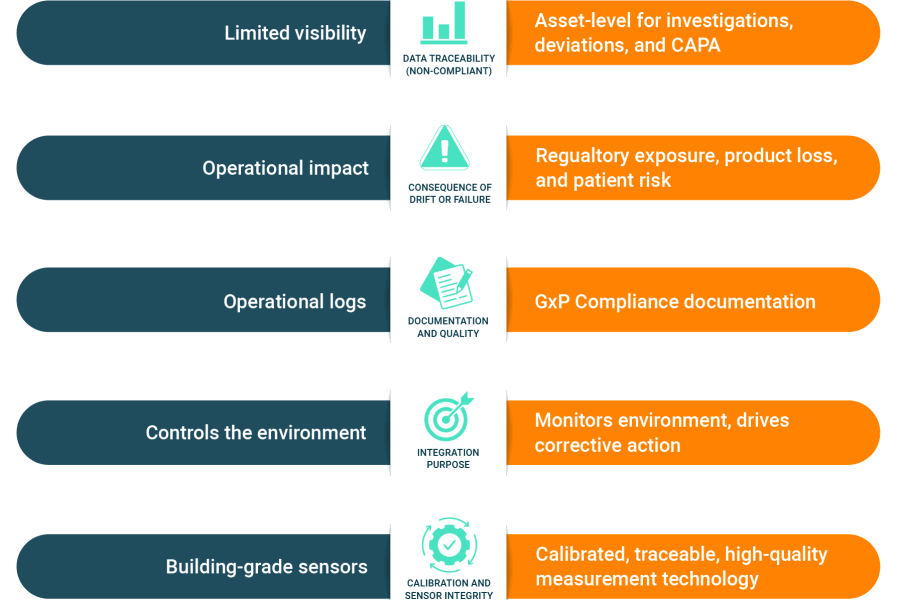
The Compliance Gap of Relying on BMS Alone
Relying on a BMS alone leaves a dangerous compliance gap. BMS data is simply not accepted as GMP-compliant evidence, often resulting in regulatory findings like FDA Form 483s and warning letters.
A BMS lacks the essential tools needed to meet global pharmaceutical standards, including:
Validated audit trails and secure electronic signatures that are required by global regulators.
Protocols to prevent data gaps or operational blind spots where critical alarms may not reach QA/QC staff.
In a worst-case scenario, this risks patient safety due to undetected or undocumented temperature or humidity excursions. Only a dedicated, validated EMS provides the necessary proof of compliance and product integrity.

The Power of Integration: EMS Driving BMS Action
In the most advanced and efficient setup, the BMS and EMS are complementary, not competing. The BMS keeps the facility running efficiently, while the EMS ensures compliance and safeguards the product. The key to unlocking their combined power is integration.
Modern Environmental Monitoring Systems, like testo Saveris 1, can seamlessly integrate with your existing BMS using webhooks and APIs. This connection creates a feedback loop that transforms monitoring data into immediate, facility-wide automation:
Triggering Corrective Actions: If EMS monitors a critical parameter and it goes out of limits, the EMS can use a webhook to automatically trigger a corrective action in the BMS (e.g., turn on an AC unit or adjust airflow). This ensures GMP limits are maintained with minimal delay.
Enabling Energy Optimization: If critical parameters are safely within limits, the EMS can signal the BMS that it is safe to optimize energy consumption. This allows the BMS to turn off or modulate AC units for energy efficiency without compromising product safety or compliance.
This integrated approach creates a unified, compliant, and responsive ecosystem where critical monitoring data not only produces audit reports but also actively drives facility automation.

Ensuring a Modernized, Compliant Facility
Building Management Systems keep facilities running. But relying on a BMS alone leaves a dangerous compliance gap.
Environmental Monitoring Systems like testo Saveris 1 ensure compliance, protect product integrity, and, ultimately, safeguard patients.
By utilizing webhook integrations and API data sharing, the EMS extends its value even further, enabling enterprise-wide data visibility and triggering BMS corrective actions.
To ensure your facility is ready for the future of pharmaceutical compliance and to guarantee audit success, choose an EMS that is built for compliance and designed for integration.

