Essential Services for Pharma Compliance
Built on calibration, validation, and environmental control

What Services Are Essential for Ongoing Pharmaceutical Compliance?
In the pharma and life sciences industries, compliance isn’t earned once and then filed away. Certification is a continuous discipline; a chain of evidence that must remain intact from calibration through validation and environmental control. The organizations leading the industry today aren’t the ones who simply “check the box.” They’re the ones who build a proactive service strategy that eliminates uncertainty.
Certification as a Lifecycle, Not a Moment
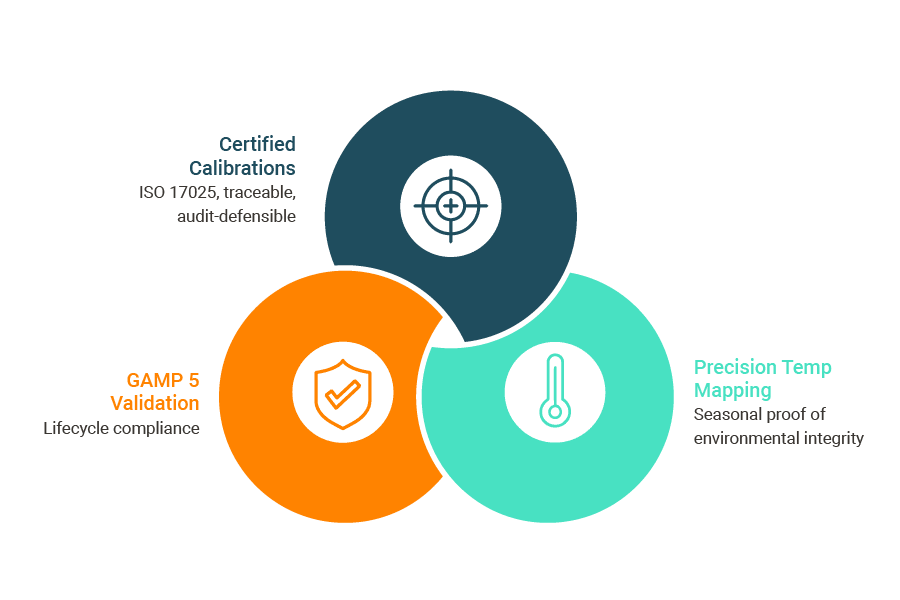
Regulatory expectations have made it clear that GxP readiness depends on three core pillars:
Calibrations must create a defensible chain of trust.
Modern pharma operations rely on ISO 17025-accredited, traceable calibrations that reduce downtime, maintain data integrity, and withstand audit scrutiny.
A system isn’t compliant until it’s validated and stays validated.
Lifecycle validation (IQ/OQ/PQ, documentation control, re-validation after changes) ensures monitoring systems remain in a state of control, not just at installation.
Environmental proof is now non-negotiable.
Your cold chain is only as strong as its weakest link, and environmental integrity is complex. Regulators require proof that your controlled areas perform under worst-case conditions. Testo’s seasonal mapping goes beyond basic verification. By conducting winter and summer validations, we proactively test the impact of systems struggling under extreme outdoor conditions. This approach demonstrates environmental fitness across the entire year, revealing subtle risks, like hot and cold spots that emerge only during peak seasons, before they lead to costly product loss or compliance failures.
Together, these pillars define what true, ongoing certification looks like in today’s regulatory landscape.
Where Testo Stands Out
While these pillars apply industry-wide, the way they’re delivered determines their impact. Testo elevates them through a U.S.-based service ecosystem designed specifically for regulated pharma environments.
Traceable, audit-ready calibrations with hot-swap options and full documentation oversight.
GAMP 5–aligned IQ/OQ/PQ, re-validation support, and clear evidence packages handled by U.S. validation experts.
Winter and summer mapping for warehouses, controlled storage areas, and cold rooms—performed by specialists who understand FDA expectations.
Local technicians deploy testo Saveris 1 with proper setup, network integration, software configuration, and structured project oversight.
On-site or virtual training, retraining, and onboarding support to ensure teams stay confident in daily operation.

The Human Advantage: Dedicated Project Managers
Every Testo client is paired with a dedicated Project Manager (PM). This single point of contact coordinates services, manages documentation, aligns stakeholders, and supports audit readiness.
Your Project Manager delivers value far beyond scheduling:
Consistent communication: One person who knows your facilities, your documentation history, and your compliance priorities equals no repeated explanations or lost context.
Fewer delays and fewer handoffs: No phone-tree frustration. No slow ticket systems. Just direct access to someone who understands what’s at stake.
Alignment across stakeholders: Whether it’s QA, facilities, IT, or corporate leadership, your PM ensures everyone is working from the same information and timeline.
This human-centered model transforms service delivery into a partnership, reducing uncertainty, strengthening audit preparedness, and helping teams shift from reactive problem-solving to continuous, predictable compliance.
From Compliance to Certainty
Certification today requires more than reliable tools. It requires a service strategy built to reduce risk, support audits, and maintain confidence year-round.
Testo’s U.S.-based calibration, validation, mapping, installation, and training services are designed to deliver exactly that, helping you build a stronger, more defensible GxP posture from day one.